One of the most common ailments modern Canadians face is inflammatory bowel disease (IBD), with an incidence rate of 1/140 Canadians—and is forecasted to increase for younger generations—making it one of the highest globally. IBD is a collective term for chronic inflammatory disorders in the small or large intestine (aka the gut) with symptoms including diarrhea, unintentional weight loss, fatigue, and abdominal pain and cramping, which debilitates patients' everyday lives.
The intestines or gut organ undergoes high regeneration compared to other human organs due to constant strain from breaking down food, absorbing nutrients, eliminating waste, and chronic inflammation in IBD cases. It does this through intestinal stem cells, which are the source of all gut cell types and require proper environmental signals to function correctly. Alterations in these signals can lead to IBD. Defining these signals and creating therapeutic strategies to advance stem cell activity in the absence of proper signalling is one of the main research aims of Dr. Tae-Hee Kim’s lab at SickKids. Indeed, the most recent publication coming from this lab addresses this by determining the as-of-yet unknown role of intestinal glial cells on regulating the neighbouring stem cells.
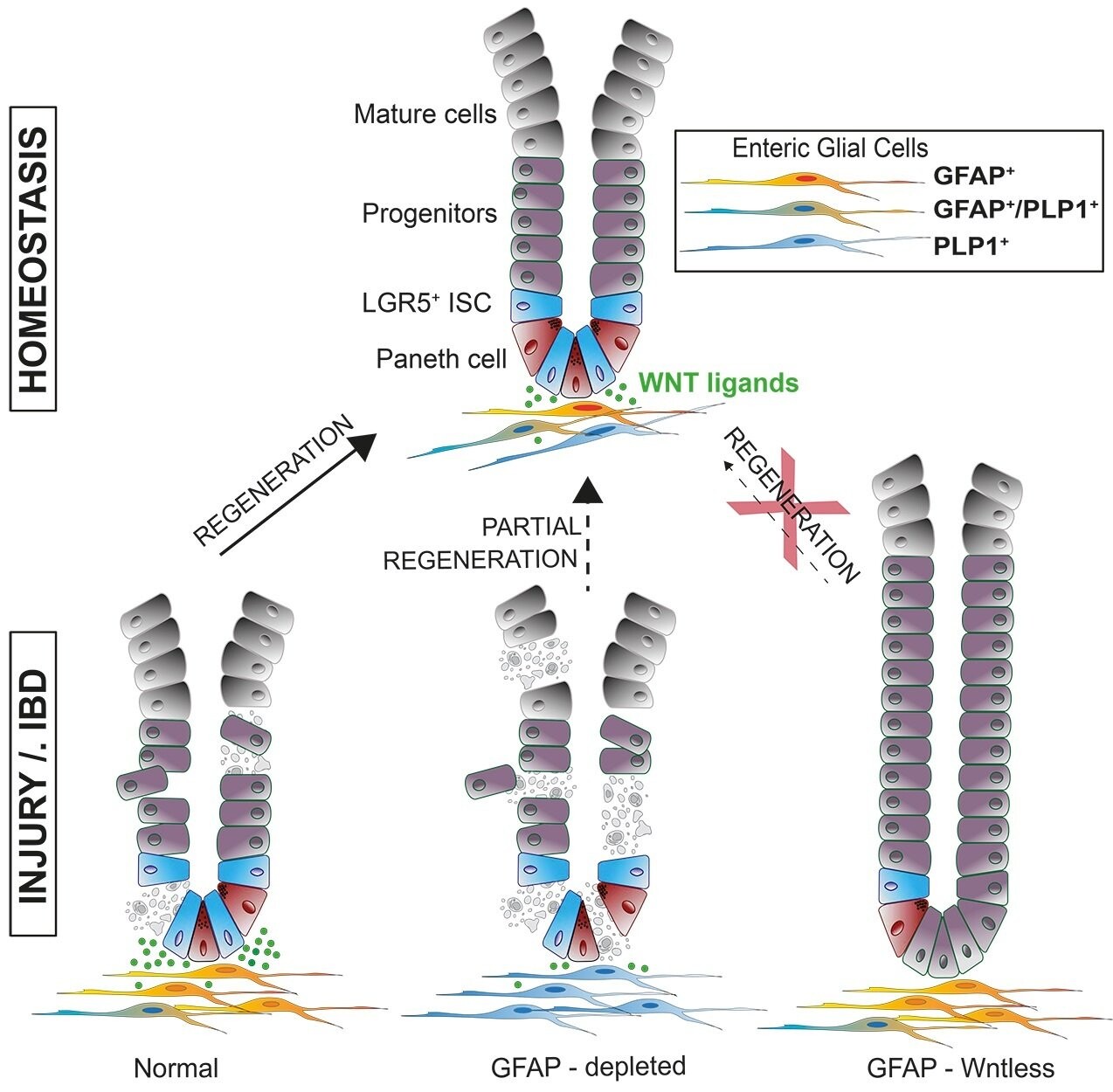
The gut is also considered the second brain of the human body due to an extensive network of hundreds of millions of two types of nervous system cells: neurons and glial cells. The glial cells outnumber neurons fourfold and are located near the intestinal stem cells, but their role as a stem cell niche has been unexplored. By analyzing human and mouse intestinal gene expression at the single-cell level and disrupting specific gene activities, alongside IBD and gut injury mouse models, Dr. Meryem Baghdadi, a postdoctoral fellow in Dr. Kim’s lab, in collaboration with Dr. Arshad Ayyaz in Dr. Jeff Wrana’s lab and other colleagues, demonstrated their essential role in intestinal maintenance.
Firstly, the group found that inactivating a specific glial cell subset decreased stem cell numbers and renewal, indicating it has a defined regulatory role in stem cell behaviour. Secondly, gut maintenance and regeneration require this cell subpopulation after damage such as IBD-induced inflammation. The team also found that this glial cell subset secretes signalling molecules, specifically WNT ligands. These molecules activate WNT signalling, an evolutionarily conserved pathway that significantly contributes to gut stem cell renewal and differentiation. This study demonstrated glial cells’ role in promoting stem cell-induced regeneration in the gut via WNT signalling through the production of ligands in response to inflammation. The mouse models further demonstrated their role in intestinal repair, where disrupted glial cell function promoted IBD symptoms such as colon damage and weight loss. In summary, the results implicate that intestinal glial cells are a crucial trigger of WNT signalling for resident stem cells to renew and promote gut repair upon injury caused by stressors such as inflammation, making it a potential role-player in IBD pathogenesis. Additionally, altering these signals potentially prevents healing of the gut, resulting in exacerbated IBD symptoms.
The results present a promising route for IBD therapeutics by modulating patient glial cells and the WNT signalling pathway to improve stem cell proliferation, potentially preventing or relieving IBD-caused inflammation and promoting intestinal repair and healing.
*For more details, read Dr. Kim’s lab’s paper titled “Enteric Glial Cell Heterogeneity Regulates Intestinal Stem Cell Niches” on Cell Stem Cell on November 1.