Small Molecule Exhibits Pan-Antiviral Activity By Adjusting The Host Cell Environment
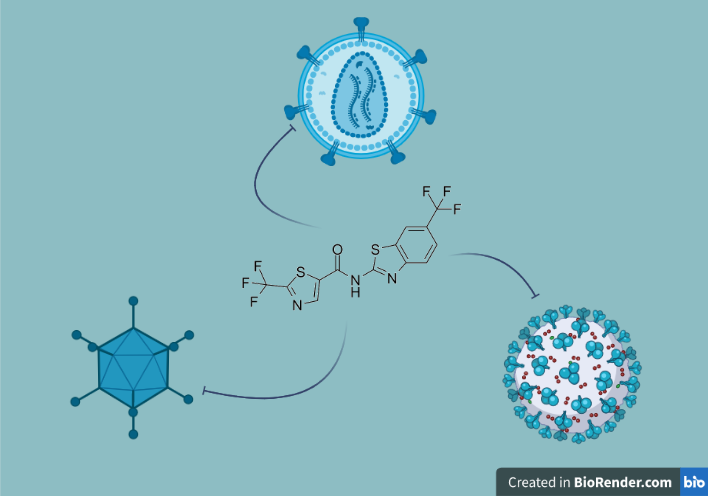
The development of antivirals traditionally focused on targeting the virus itself. The majority of these drugs aim at viral enzymes, such as proteases and polymerases, and entry proteins used for interacting with their host cell receptor, resulting in them being effective against only a narrow spectrum of viruses. Although these drugs succeed in reducing infections in the short term, rapid viral evolution and mutation rates often sprout drug-resistant strains. In a recent publication out of Dr. Alan Cochrane’s research group, first author Subha Dahal and others investigated an alternative antiviral strategy, using a single compound to combat multiple viruses by modulating the cell environment and rendering it unfavourable for viral replication or reproduction. The lab primarily specializes in identifying host factors that regulate HIV-1 RNA processing and characterizing small molecules that target this process. However, in response to the current COVID-19 pandemic, the Cochrane lab, alongside Dr. Walid A. Houry’s group at UofT's Department of Biochemistry, broadened their research interests to identify novel inhibitors of coronaviruses. The research organized here demonstrates how critical interdisciplinary collaboration between multiple labs and institutes can be, facilitating this research focus shift and the sharing of expertise and established resources at the University of Toronto. The discovery that one compound can inhibit a diverse range of viruses illustrates the promise of host-directed therapeutics and their use against emerging infections.
Previous research and concept behind host-directed viral therapeutics
“Viruses, as parasites, are dependent on many host cell functions for their replication, so our rationale is that many different viruses could use the same host cell functions/pathways to reproduce. If we can target these critical processes, we could develop an agent effective against multiple different viruses,” stated Dr. Cochrane. These drugs would tweak the cellular environment so that the host would not support virus replication or new virus formation. Dahal noted, “Since the therapeutic is host-directed, the virus will have a harder time developing drug resistance as the drug forces it to exploit an entirely new pathway to overcome the activity of the antiviral”. An instance of an essential host pathway is alternative RNA splicing, which various mammal-infecting viruses depend on for reproduction, with the prime examples being HIV and adenovirus. The main challenge with this therapeutic strategy, as Dahal stated, is identifying the proper dosage, high enough to inhibit the virus without any adverse effects on host cells.
Previous work from the group identified an organic compound stilbene 2-(2-(5-nitro-2-thienyl)vinyl)quinoline that modifies viral RNA accumulation and inhibits HIV-1 replication. Building on this finding, the team’s organic chemist Dr. David Grierson from the University of British Columbia, synthesized and screened a library of structural variants, eventually selecting a thiazole amide compound designated as GPS491. In their latest publication in the open-access MDPI journal Viruses, the research group uncovered broad-spectrum activity of GPS491 against three classes of unrelated viruses that infect humans-HIV, adenovirus, and coronaviruses. They show that GPS491 inhibits HIV-1 protein expression partially by altering viral RNA accumulation and processing. They also observed similar changes in viral RNA processing with human adenovirus. Surprisingly, the same compound also proved effective against multiple human coronaviruses, two seasonal common cold strains (HCov-229E and HCov-OC43) and pathogenic SARS-CoV-2. In short, the group showcases the pan-antiviral activity of GPS491, implicating that this compound blocks viruses that have entirely different life cycles: HIV and adenovirus, which both replicate in the nucleus, while coronaviruses reproduce only in the cell cytoplasm.
GPS491 Activity Against HIV-1
The group assessed whether GPS491 could inhibit replication of multiple HIV-1 strains, the predominant HIV type. They found that the compound suppressed all the HIV-1 strains, including wild-type and antiretroviral drug-resistant ones. It’s important to note that the treatment was potent even after the virus integrated into the host genome, indicating the compound acts after cell entry and infection. Additional experiments revealed that low GPS491 doses reduced HIV-1 protein levels by over 90% and altered the levels of specific HIV-1 RNAs. These results suggest that the compound mainly targets the virus's RNA processing. The effectiveness against HIV strains resistant to currently available drugs holds the most relevance, showing the promise of using these compounds as a complementary treatment in a medical or health setting.
GPS491 Activity Against Adenoviruses
In addition to HIV, the group tested GPS491 on adenoviruses—which have double-stranded DNA genomes compared to RNA genomes of HIV and coronaviruses—with aid and resources from fellow Department of Molecular Genetics faculty member Dr. Martha Brown’s group. Both labs previously collaborated on testing the efficacy of cardiotonic steroids in treating HIV and adenoviruses. They demonstrated that adding GPS491 to cell culture inoculated with human adenovirus-C5 reduced viral yield 1000-fold. Experiments showcased a reduced expression of the viral structural proteins—hexon and E1A—after adding GPS491. The researchers then analyzed adenovirus gene expression and found that GPS491 reduced the abundance of most viral RNAs and altered E1A RNA splicing over the course of infection, indicating that GPS491 alters RNA processing in adenoviruses as well.
GPS491 Activity Against Coronaviruses
With the emergence and continuation of the COVID-19 pandemic, the Cochrane lab incorporated coronaviruses in their research. The viral core of UofT’s C-CL3 unit, headed by one of our Department’s faculty members Dr. Scott Gray-Owen and other notable UofT PIs, facilitated the testing of GPS491’s efficacy against the coronavirus family, which has a fundamentally different replication cycle compared to HIV and adenovirus. They specifically examined human coronaviruses 229E and OC43, which cause mild or moderate respiratory illness akin to the cold, and SARS-CoV-2, the agent behind the current pandemic. Despite the differences among these viruses, GPS491 inhibited the replication of all three coronaviruses replication indicated by the loss of viral RNA accumulation and nucleocapsid (N) and spike (S) protein expression. GPS491 retained its antiviral activity even after the virus enters the cells and initiates new virus formation, indicating that the compound impinges upon a critical stage in the viral life cycle when it already takes up residence in its host cell. SARS-CoV-2 is only the latest of several pathogenic coronaviruses, alongside SARS-CoV-1 and MERS, that have emerged, making it likely that novel members of the coronavirus family will arise in the future. Therefore, a drug effective against all coronavirus species would be invaluable.
GPS491 Activity Against Host Cell Proteins
The group observed that GPS491 treatment alters the abundance and phosphorylation of specific members of the SR protein family, part of the host cellular proteome involved in regulating RNA splicing and processing. Notably, these proteins are heavily involved in regulating gene expression and replication of HIV-1. One noteworthy finding was the compound’s inhibition of SRSF10, a key player in regulating HIV-1 and hepatitis B replication, which resulted in modifying the alternative splicing of the Bcl-x gene, involved in regulating apoptosis or cell death. Previous studies determined that altered SR proteins expression impacts adenovirus replication and alternative splicing and the replication of other RNA viruses, including SARS-CoV-1/2. In short, SR proteins are crucial components in the replication of multiple viruses, those that require RNA splicing such as HIV and adenovirus and that don’t splice such as coronaviruses—and are a potential target of GPS491.
Conclusion and Future Research
Overall, this study unearthed a promising pan-antiviral in GPS491. The results display the compound’s ability to inhibit replication cycles of several unrelated viruses that infect humans—retroviruses (the family of HIV), adenoviruses and coronaviruses—by targeting host cell proteins involved in RNA processing with minimal damage to the host. Moreover, the results indicate the compound functions after the virus enters and infects its host cell. Future studies will aim to elucidate the detailed mechanism of action of GPS491 to fully realize its potential.
The lab also has several other potential pan-antiviral compounds in the testing pipeline. Animal testing in mice is ongoing to examine toxicity and efficacy in the C-CL3 unit, in collaboration with the biotech company ViroCarb Inc. “The primary focus now is a combination of identifying the compounds and elucidating their mechanism of action” stated Dr. Cochrane, “that would allow us to enhance the antiviral effects and reduce the off-target or negative effects of the compound on the host”. He also stressed that although COVID-19 vaccines were developed relatively quickly and have high efficacy rates, there is no guarantee of similar success against future emerging infections, as demonstrated by the lack of an HIV vaccine. As a result, having broad-spectrum antivirals would be beneficial to have on hand in response to any prospective epidemic or pandemic, especially in the early, pre-vaccine development stages. For that to happen, interdisciplinary and cross-institutional research will be vital going into the future.
A big thank you to Dr. Alan Cochrane and first author Subha Dahal for offering their insight and expertise in this piece!


