Main Second Level Navigation
Oct 7, 2022
Study of intronic variant provides critical information about the size and growth speed of gliomas
Research Highlights
MoGen scientists stride towards a better understanding of the biology of non-coding genomic alterations
Daniel Schramek
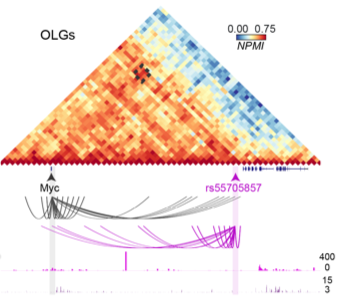
Heat map showing the interaction between the Myc gene and the rs55705857 locus in mouse oligodendrocytes and their precursor cells (OLGs) in the somatosensory cortex of mice.